ADCETRIS® Clinical Studies
(brentuximab vedotin)
14 CLINICAL STUDIES
14.1 Classical Hodgkin Lymphoma
Randomized Clinical Trial in Previously Untreated Stage III or IV Classical Hodgkin Lymphoma (Study 5: ECHELON-1, NCT01712490)
The efficacy of ADCETRIS in combination with chemotherapy for the treatment of patients with previously untreated Stage III or IV cHL was evaluated in a randomized, open-label, 2-arm, multicenter trial. Of the 1334 total patients, 664 patients were randomized to the ADCETRIS + doxorubicin [A], vinblastine [V] and dacarbazine [D] (ADCETRIS + AVD) arm and 670 patients were randomized to the A+ bleomycin [B] + V + D (ABVD) arm. Patients in both treatment arms were treated intravenously on Days 1 and 15 of each 28-day cycle for up to 6 cycles. Dosing in each treatment arm was administered according to the following:
- •
- ADCETRIS + AVD arm: ADCETRIS 1.2 mg/kg over 30 minutes, doxorubicin 25 mg/m2, vinblastine 6 mg/m2, and dacarbazine 375 mg/m2
- •
- ABVD arm: doxorubicin 25 mg/m2, bleomycin 10 units/m2, vinblastine 6 mg/m2, and dacarbazine 375 mg/m2
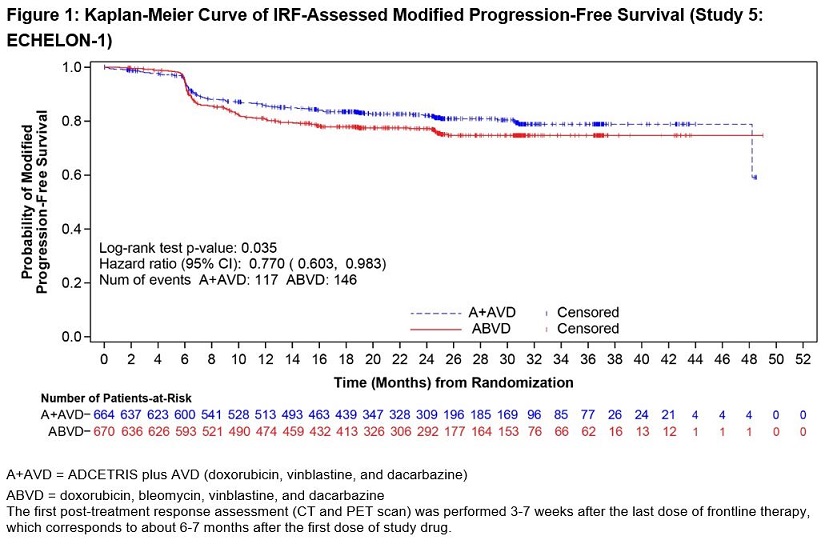
Efficacy was established based on modified progression-free survival (modified PFS) per independent review facility (IRF). A modified PFS event is defined as progression, death, or receipt of additional anticancer therapy for patients who are not in a complete response (CR) after completion of frontline therapy.
Patients had Stage III (36%) or IV disease (64%), and 62% had extranodal involvement at diagnosis. Most patients were male (58%) and white (84%). The median age was 36 years (range, 18‑83); 186 patients (14%) were 60 years or older.
The efficacy results are summarized in Table 13 and Figure 1.
| ||
Modified Progression-Free Survival per IRF* | ADCETRIS + AVD | ABVD |
Number of events (%) | 117 (18%) | 146 (22%) |
Median months (95% CI) | NE† | NE† |
Hazard ratio (95% CI)‡ | 0.77 (0.60, 0.98) | |
P-value§ | 0.035 | |
Reason leading to a modified PFS event | ||
Progressive disease | 90 (14) | 102 (15) |
Death due to any cause | 18 (3) | 22 (3) |
Receipt of additional anticancer therapy for | 9 (1) | 22 (3) |
At the time of the modified PFS analysis, the prespecified interim OS analysis did not demonstrate a significant difference. The CR rate per IRF assessment at the end of the randomized regimen was 73% on the ADCETRIS + AVD arm and 70% on the ABVD arm.
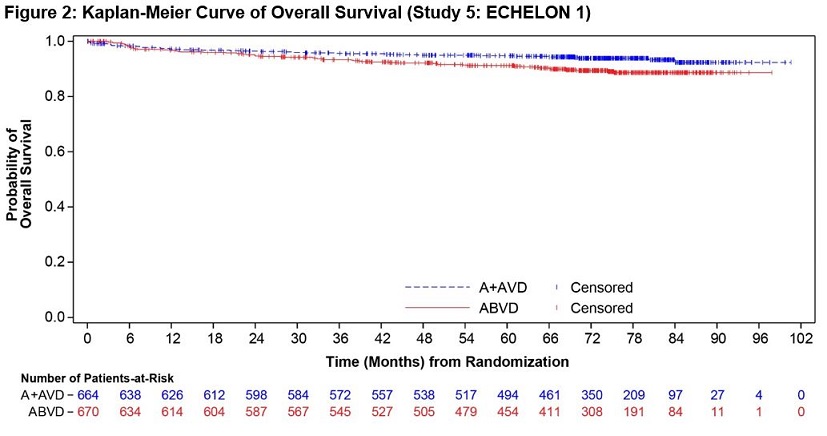
A prespecified second interim analysis showed a statistically significant improvement in OS in the ADCETRIS + AVD arm (39 deaths) compared to the ABVD arm (64 deaths). With an estimated median follow-up of 6.1 years, the stratified hazard ratio was 0.59 (95% CI, 0.396; 0.879), with a 2-sided p-value of 0.009 (significance level, 0.0365). Median OS was not reached in either treatment arm (Figure 2).
Randomized Clinical Trial in Previously Untreated High Risk Classical Hodgkin Lymphoma (Study 7, AHOD1331, NCT02166463)
The efficacy of ADCETRIS in combination with chemotherapy for the treatment of pediatric patients (2 to <22 years of age) with previously untreated, high risk cHL was evaluated in a randomized, open-label, actively controlled trial. High risk was defined as Ann Arbor Stage IIB with bulk disease, Stage IIIB, Stage IVA, and Stage IVB. Of the 600 total patients randomized, 300 were randomized to ADCETRIS + Doxorubicin [A], Vincristine [V], Etoposide [E], Prednisone [P], Cyclophosphamide [C] (ADCETRIS + AVEPC) arm and 300 patients were randomized to A+ Bleomycin [B]+V+E+P+C (ABVE-PC) arm. Patients in each treatment arm received up to 5 cycles of the following:
- •
- ADCETRIS + AVEPC arm: ADCETRIS 1.8 mg/kg over 30 minutes (day 1), doxorubicin 25 mg/m2 (days 1 and 2), vincristine 1.4 mg/m2 (day 8), etoposide 125 mg/m2 (days 1-3), prednisone 20 mg/m2 BID (days 1-7), cyclophosphamide 600 mg/m2 (days 1 and 2)
- •
- ABVE-PC arm: doxorubicin 25 mg/m2 (days 1 and 2), bleomycin 5 units/m2 (day1) and 10 units/m2 (day 8), vincristine 1.4 mg/m2 (days 1 and 8), etoposide 125 mg/m2 (days 1-3), prednisone 20 mg/m2 BID (days 1-7), cyclophosphamide 600 mg/m2 (days 1 and 2)
The median age was 15 years (range: 3-21 years); 53% were male, 74% were White, 11% Black, and 3% Asian. Nine patients were <6 years, 81 patients were 6 to <12 years, 448 patients were 12 to <18 years, and 62 patients were ≥18 years. Of the 600 enrolled patients, 20% had disease stage of IIB with bulk disease, 19% had IIIB, 29% had IVA, and 31% had IVB.
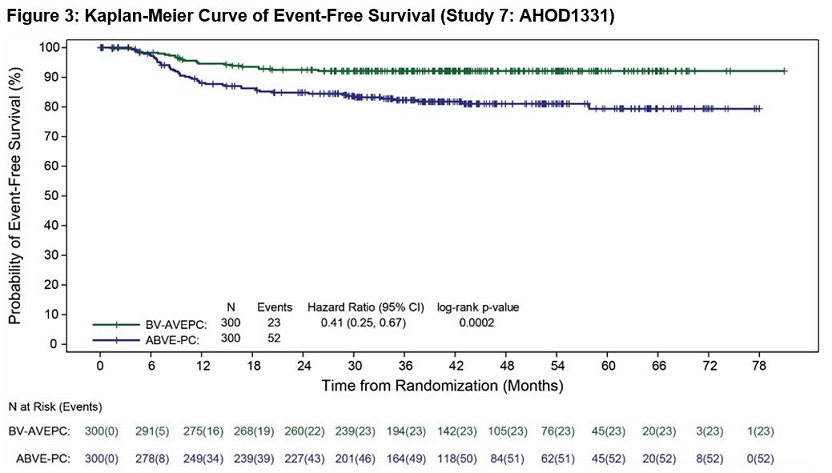
Efficacy was established based on event-free-survival (EFS), defined as the time from randomization to the earliest of disease progression or relapse, second malignancy, or death due to any cause. Efficacy results are summarized in Table 14.
| NR = Not reached. | ||
| ||
Event-Free Survival | ADCETRIS + AVEPC | ABVE-PC |
Number of Events (%) | 23 (8) | 52 (17) |
Median (95% CI) | NR | NR |
Hazard Ratio (95% CI)* | 0.41 (0.25, 0.67) | |
P-value (log-rank test)† | 0.0002 | |
Randomized Placebo-Controlled Clinical Trial in Classical Hodgkin Lymphoma Post-Auto-HSCT Consolidation (Study 3: AETHERA, NCT01100502)
The efficacy of ADCETRIS in patients with cHL at high risk of relapse or disease progression post-auto-HSCT was studied in a randomized, double-blind, placebo-controlled clinical trial. Three hundred twenty-nine (329) patients were randomized 1:1 to receive placebo or ADCETRIS 1.8 mg/kg intravenously over 30 minutes every 3 weeks for up to 16 cycles, beginning 30–45 days post-auto-HSCT. Patients in the placebo arm with progressive disease per investigator could receive ADCETRIS as part of a separate trial. The primary endpoint was progression-free survival (PFS) determined by independent review facility (IRF). Standard international guidelines were followed for infection prophylaxis for HSV, VZV, and PJP post-auto-HSCT [see Clinical Trial Experience (6.1)].
High risk of post-auto-HSCT relapse or progression was defined according to status following frontline therapy: refractory, relapse within 12 months, or relapse ≥12 months with extranodal disease. Patients were required to have obtained a complete response (CR), partial response (PR), or stable disease (SD) to most recent pre-auto-HSCT salvage therapy.
A total of 329 patients were enrolled and randomized (165 ADCETRIS, 164 placebo); 327 patients received study treatment. Patient demographics and baseline characteristics were generally balanced between treatment arms. The 329 patients ranged in age from 18–76 years (median, 32 years) and most were male (53%) and white (94%). Patients had received a median of 2 prior systemic therapies (range, 2–8) excluding autologous hematopoietic stem cell transplantation.
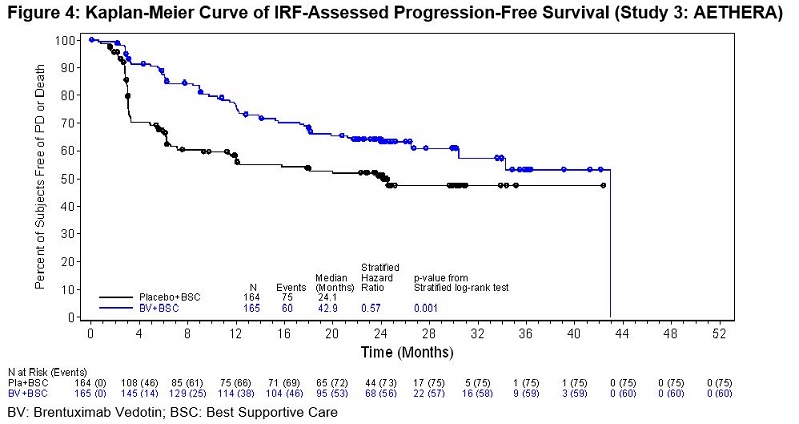
The efficacy results are summarized in Table 15. PFS is calculated from randomization to date of disease progression or death (due to any cause). The median PFS follow-up time from randomization was 22 months (range, 0–49). Study 3 (AETHERA) demonstrated a statistically significant improvement in IRF-assessed PFS and increase in median PFS in the ADCETRIS arm compared with the placebo arm. At the time of the PFS analysis, an interim overall survival analysis demonstrated no difference.
Progression-Free Survival per IRF | ADCETRIS | Placebo |
Number of events (%) | 60 (36) | 75 (46) |
Median months (95% CI) | 24.1 (11.5, NE†) | |
Stratified Hazard Ratio (95% CI) | 0.57 (0.40, 0.81) | |
Stratified Log-Rank Test P-value | 0.001 | |
Clinical Trial in Relapsed Classical Hodgkin Lymphoma (Study 1, NCT00848926)
The efficacy of ADCETRIS in patients with cHL who relapsed after autologous hematopoietic stem cell transplantation was evaluated in one open-label, single-arm, multicenter trial. One hundred two (102) patients were treated with 1.8 mg/kg of ADCETRIS intravenously over 30 minutes every 3 weeks. An independent review facility (IRF) performed efficacy evaluations which included overall response rate (ORR = complete response [CR] + partial response [PR]) and duration of response as defined by clinical and radiographic measures including computed tomography (CT) and positron-emission tomography (PET) as defined in the 2007 Revised Response Criteria for Malignant Lymphoma (modified).
The 102 patients ranged in age from 15–77 years (median, 31 years) and most were female (53%) and white (87%). Patients had received a median of 5 prior therapies including autologous hematopoietic stem cell transplantation.
The efficacy results are summarized in Table 16. Duration of response is calculated from date of first response to date of progression or data cutoff date.
N = 102 | |||
Percent (95% CI) | Duration of Response, in months | ||
Median (95% CI) | Range | ||
CR | 32 (23, 42) | 20.5 (12.0, NE*) | 1.4 to 21.9† |
PR | 40 (32, 49) | 3.5 (2.2, 4.1) | 1.3 to 18.7 |
ORR | 73 (65, 83) | 6.7 (4.0, 14.8) | 1.3 to 21.9† |
14.2 Systemic Anaplastic Large Cell Lymphoma and Other CD30-Expressing Peripheral T-Cell Lymphomas
Randomized Clinical Trial in Previously Untreated Systemic Anaplastic Large Cell Lymphoma or Other CD30-Expressing Peripheral T-Cell Lymphomas (Study 6: ECHELON-2, NCT01777152)
The efficacy of ADCETRIS in combination with chemotherapy for the treatment of adult patients with previously untreated, CD30-expressing PTCL was evaluated in a multicenter, randomized, double-blind, double-dummy, actively controlled trial. For enrollment, the trial required CD30 expression ≥10% per immunohistochemistry. The trial excluded patients with primary cutaneous CD30-positive T-cell lymphoproliferative disorders and lymphomas. The trial required hepatic transaminases ≤3 times ULN, total bilirubin ≤1.5 times ULN, and serum creatinine ≤2 times ULN.
Of the 452 total patients, 226 patients were randomized to the ADCETRIS + CHP arm and 226 patients were randomized to the CHOP arm. Patients in both treatment arms were treated intravenously on Day 1 of each 21-day cycle for 6 to 8 cycles; prednisone was administered orally on Days 1‑5. Dosing in each treatment arm was administered according to the following:
- •
- ADCETRIS + CHP arm: ADCETRIS 1.8 mg/kg over 30 minutes, cyclophosphamide 750 mg/m2, doxorubicin 50 mg/m2, and prednisone 100 mg orally
- •
- CHOP arm: cyclophosphamide 750 mg/m2, doxorubicin 50 mg/m2, vincristine 1.4 mg/m2, and prednisone 100 mg orally
The median age was 58 years (range: 18 to 85), 63% were male, 62% were White, 22% were Asian, and 78% had an ECOG performance status of 0-1. Of the 452 patients enrolled, the disease subtypes included patients with systemic ALCL [70%; 48% anaplastic lymphoma kinase (ALK) negative and 22% ALK positive], PTCL NOS (16%), angioimmunoblastic T-cell lymphoma (12%), adult T-cell leukemia/lymphoma (2%), and enteropathy-associated T-cell lymphoma (<1%). Most patients had Stage III or IV disease (81%) and a baseline international prognostic index of 2 or 3 (63%).
During randomized treatment, on the ADCETRIS + CHP arm, 70% of patients received 6 cycles and 18% of patients received 8 cycles. On the CHOP arm, 62% of patients received 6 cycles and 19% received 8 cycles.
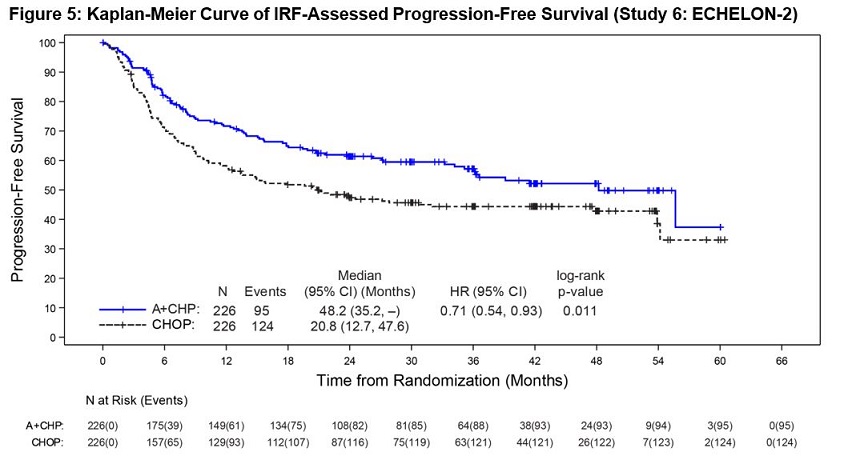
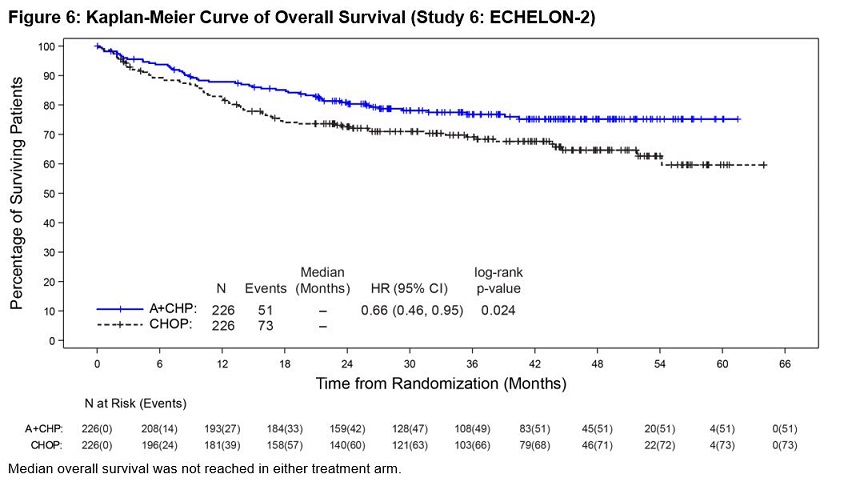
Efficacy was based on IRF-assessed PFS, which was defined as time from randomization to progression, death due to any cause, or receipt of subsequent anticancer chemotherapy to treat residual or progressive disease. Other efficacy endpoints included PFS in patients with systemic ALCL, overall survival, complete response rate, and overall response rate. Efficacy results are summarized in Table 17. Kaplan-Meier curves for PFS and overall survival are presented in Figure 5 and Figure 6, respectively.
| NE = Not estimable | ||
| ||
Outcomes per IRF* | ADCETRIS + CHP | CHOP |
PFS | ||
Number of events, n (%) | 95 (42) | 124 (55) |
Median PFS, months (95% CI) | 48.2 (35.2, NE) | 20.8 (12.7, 47.6) |
Hazard ratio (95% CI)† | 0.71 (0.54, 0.93) | |
P-value‡ | 0.011 | |
Reason leading to a PFS event, n (%) | ||
Progressive disease | 71 (31) | 86 (38) |
Death | 13 (6) | 17 (8) |
Receipt of subsequent anticancer chemotherapy to treat residual or progressive disease | 11 (5) | 21 (9) |
PFS for patients with sALCL | ||
N | 163 | 151 |
Number of patients with a PFS event, n (%) | 56 (34) | 73 (48) |
Median PFS, months (95% CI) | 55.7 (48.2, NE) | 54.2 (13.4, NE) |
Hazard ratio (95% CI)† | 0.59 (0.42, 0.84) | |
P-value‡ | 0.003 | |
OS§ | ||
Number of deaths | 51 (23) | 73 (32) |
Median OS, months (95% CI) | NE (NE, NE) | NE (54.2, NE) |
Hazard ratio (95% CI)† | 0.66 (0.46, 0.95) | |
P-value‡ | 0.024 | |
CR Rate¶ | ||
% (95% CI) | 68 (61, 74) | 56 (49, 62) |
P-value# | 0.007 | |
ORR¶ | ||
% (95% CI) | 83 (78, 88) | 72 (66, 78) |
P-value# | 0.003 | |
14.3 Systemic Anaplastic Large Cell Lymphoma
Clinical Trial in Relapsed sALCL (Study 2, NCT00866047)
The efficacy of ADCETRIS in patients with relapsed sALCL was evaluated in one open-label, single-arm, multicenter trial. This trial included patients who had sALCL that was relapsed after prior therapy. Fifty-eight (58) patients were treated with 1.8 mg/kg of ADCETRIS administered intravenously over 30 minutes every 3 weeks. An IRF performed efficacy evaluations which included overall response rate (ORR = complete response [CR] + partial response [PR]) and duration of response as defined by clinical and radiographic measures including computed tomography (CT) and positron-emission tomography (PET) as defined in the 2007 Revised Response Criteria for Malignant Lymphoma (modified).
The 58 patients ranged in age from 14–76 years (median, 52 years) and most were male (57%) and white (83%). Patients had received a median of 2 prior therapies; 26% of patients had received prior autologous hematopoietic stem cell transplantation. Fifty percent (50%) of patients were relapsed, and 50% of patients were refractory to their most recent prior therapy. Seventy-two percent (72%) were anaplastic lymphoma kinase (ALK)-negative.
The efficacy results are summarized in Table 18. Duration of response is calculated from date of first response to date of progression or data cutoff date.
| N = 58 | ||
Percent (95% CI) | Duration of Response, in months | ||
Median (95% CI) | Range | ||
CR | 57 (44, 70) | 13.2 (10.8, NE*) | 0.7 to 15.9† |
PR | 29 (18, 41) | 2.1 (1.3, 5.7) | 0.1 to 15.8† |
ORR | 86 (77, 95) | 12.6 (5.7, NE*) | 0.1 to 15.9† |
14.4 Primary Cutaneous Anaplastic Large Cell Lymphoma and CD30-Expressing Mycosis Fungoides
Randomized Clinical Trial in Primary Cutaneous Anaplastic Large Cell Lymphoma and CD30-expressing Mycosis Fungoides (Study 4: ALCANZA, NCT01578499)
The efficacy of ADCETRIS in patients with primary cutaneous anaplastic large cell lymphoma (pcALCL) or mycosis fungoides (MF) requiring systemic therapy was studied in ALCANZA, a randomized, open-label, multicenter clinical trial. In ALCANZA, one hundred thirty-one (131) patients were randomized 1:1 to receive ADCETRIS 1.8 mg/kg intravenously over 30 minutes every 3 weeks or physician’s choice of methotrexate (5 to 50 mg orally weekly) or bexarotene (300 mg/m2 orally daily). The randomization was stratified by baseline disease diagnosis (MF or pcALCL). Patients could receive a maximum of 16 cycles (21-day cycle) of therapy every 3 weeks for those receiving brentuximab vedotin or 48 weeks of therapy for those in the control arm.
Patients with pcALCL must have received prior radiation or systemic therapy, and must have at least 1 biopsy with CD30-expression of ≥10%. Patients with MF must have received prior systemic therapy and have had skin biopsies from at least 2 separate lesions, with CD30-expression of ≥10% in at least 1 biopsy.
A total of 131 patients were randomized (66 ADCETRIS, 65 physician’s choice). The efficacy results were based on 128 patients (64 patients in each arm with CD30-expression of ≥10% in at least one biopsy). Among 128 patients, the patients’ age ranged from 22–83 years (median, 60 years), and 55% of them were male and 85% of them were white. Patients had received a median of 4 prior therapies (range, 0–15), including a median of 1 prior skin-directed therapy (range, 0–9) and 2 systemic therapies (range, 0–11). At study entry, patients were diagnosed as Stage 1 (25%), Stage 2 (38%), Stage 3 (5%), or Stage 4 (13%).
Efficacy was established based on the proportion of patients achieving an objective response (CR+PR) that lasts at least 4 months (ORR4). ORR4 was determined by independent review facility (IRF) using the global response score (GRS), consisting of skin evaluations per modified severity-weighted assessment tool (mSWAT), nodal and visceral radiographic assessment, and detection of circulating Sézary cells (MF patients only). Additional efficacy outcome measures included proportion of patients achieving a complete response (CR) per IRF, and progression-free survival (PFS) per IRF.
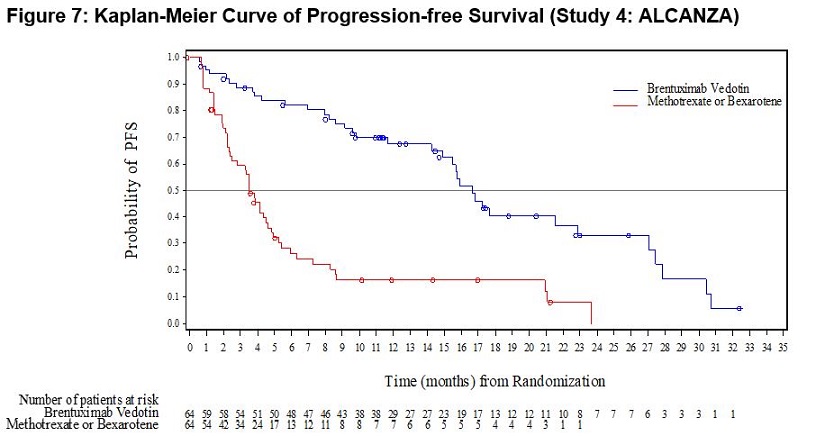
The efficacy results are summarized in Table 19 below and the Kaplan-Meier curves of IRF-assessed PFS are shown in Figure 7.
| ||
ADCETRIS | Physician’s Choice* | |
ORR4† | ||
Percent (95% CI‡) | 56.3 (44.1, 68.4) | 12.5 (4.4, 20.6) |
P-value§ | <0.001 | |
ORR | 67.2 (55.7, 78.7) | 20.3 (10.5, 30.2) |
CR | ||
Percent (95% CI‡) | 15.6 (7.8, 26.9) | 1.6 (0, 8.4) |
0.0066 | ||
PR | 51.6 (39.3, 63.8) | 18.8 (9.2, 28.3) |
PFS | ||
Number of events (%) | 36 (56.3) | 50 (78.1) |
Median months (95% CI‡) | 16.7 (14.9, 22.8) | 3.5 (2.4, 4.6) |
Hazard Ratio (95% CI‡) | 0.27 (0.17, 0.43) | |
<0.001 | ||
Supportive trials include 2 single-arm trials, which enrolled patients with MF who were treated with ADCETRIS 1.8 mg/kg intravenously over 30 minutes every 3 weeks. Out of 73 patients with MF from the 2 pooled supportive trials, 34% (25/73) achieved ORR4. Among these 73 patients, 35 had 1% to 9% CD30-expression and 31% (11/35) achieved ORR4.
14.5 Relapsed or Refractory Large B-Cell Lymphoma (LBCL)
Randomized Clinical Trial in Relapsed or Refractory Large B-Cell Lymphoma (Study 8: ECHELON-3)
The efficacy of ADCETRIS in combination with lenalidomide and a rituximab product for the treatment of adult patients with relapsed or refractory LBCL after two or more lines of systemic therapy was evaluated in ECHELON-3 (NCT04404283), a randomized, multicenter, double-blind, placebo-controlled trial. Eligible patients were 18 years of age and older with relapsed or refractory LBCL, including DLBCL NOS, DLBCL arising from indolent lymphoma, or HGBL, an ECOG performance status of 0–2, and were not eligible to receive an auto-HSCT or CAR T-cell therapy. The study excluded patients with active CNS lymphoma or Grade 2 or higher peripheral neuropathy.
Patients were randomized 1:1 to receive ADCETRIS plus lenalidomide and a rituximab product or to receive placebo plus lenalidomide and a rituximab product until disease progression or unacceptable toxicity. Randomization was stratified by CD30 expression (≥1% versus <1%), prior allogenic or autologous HSCT therapy, prior CAR T-cell therapy and cell of origin (germinal-center B-cell like [GCB] or non-GCB). Dosing in each treatment arms as follows:
- •
- ADCETRIS plus lenalidomide and rituximab arm: ADCETRIS 1.2 mg/kg via intravenous infusion every 3 weeks, lenalidomide 20 mg orally daily, and rituximab 375 mg/m2 via intravenous infusion every 3 weeks. Starting with Cycle 2, rituximab intravenous treatment could be substituted with rituximab 1,400 mg and hyaluronidase human 23,400 Units via subcutaneous injection every 3 weeks.
- •
- Placebo plus lenalidomide and rituximab arm: lenalidomide 20 mg orally daily, and rituximab 375 mg/m2 via intravenous infusion every 3 weeks. Starting with Cycle 2, rituximab intravenous treatment could be substituted with rituximab 1,400 mg and hyaluronidase human 23,400 Units via subcutaneous injection every 3 weeks.
Prophylaxis with granulocyte-colony stimulating factor (G-CSF) was mandated for both arms.
Of the 230 patients randomized (112 to ADCETRIS plus lenalidomide and rituximab, 118 to placebo plus lenalidomide and rituximab), the median age was 71 years (range 21 to 89 years); 57% were male, 53% were White, 26% were Asian and 4% were Hispanic or Latino. Race and ethnicity was not reported in 46 (20%) patients. There were no Black or African American patients enrolled. Of the total 230 patients, 71% had DLBCL NOS, 26% had transformed disease from prior indolent lymphoma, 16% had HGBL with MYC and BCL2 and/or BCL6 rearrangements or HGBL NOS. Of the patients with DLBCL NOS, 73 (45%) and 91 (55%) had GCB and non-GCB subtypes, respectively. Twenty-nine percent of patients had received prior CAR T-cell therapy and 12% had received prior HSCT. The median number of prior systemic therapies was 3 (range 2-8) with 41% receiving 2 prior therapies and 50% receiving 3 or more prior therapies. Eighty four percent had refractory disease to last therapy.
The major efficacy outcome was overall survival (OS). Additional efficacy outcome measures included progression free survival (PFS) and Objective Response Rate (ORR) according to investigator assessment per the Lugano Criteria for Response Assessment 2014. The results are presented in Table 20 and Figures 8 and 9.
| CI = confidence interval; CR = complete response; HR = hazard ratio; PFS = progression free survival; OS = overall survival. Stratified analyses were based on the stratification factors of cell of origin (GCB, non-GCB) and CD30 status (≥1%, <1%) at randomization. | ||
| ||
ADCETRIS + Lenalidomide + Rituximab (N=112) | Placebo + Lenalidomide + Rituximab (N=118) | |
Overall Survival | ||
Deaths, n (%) | 58 (51.8) | 76 (64.4) |
Median, months (95% CI) | 13.8 (10.3, 18.8) | 8.5 (5.4, 11.7) |
HR (95% CI)* | 0.63 (0.45, 0.89) | |
0.0085 | ||
PFS per Investigator | ||
Number (%) of patients with event | 71 (63.4) | 85 (72.0) |
Progressive disease, n (%) | 56 (50.0) | 71 (60.2) |
Death, n (%) | 15 (13.4) | 14 (11.9) |
Median, months (95% CI) | 4.2 (2.9, 7.1) | 2.6 (1.4, 3.1) |
HR (95% CI)* | 0.53 (0.38, 0.73) | |
p-value† | <0.0001 | |
Objective Response Rate per Investigator | ||
Objective response rate (CR or PR), n (%) | 72 (64.3) | 49 (41.5) |
95% CI for ORR§ | (54.7, 73.1) | (32.5, 51.0) |
p-value¶ | 0.0006 | |
Complete response (CR), n (%) | 45 (40.2) | 22 (18.6) |
Figure 8: Kaplan-Meier Curve of Overall Survival (Study 8: ECHELON-3)
Figure 9: Kaplan-Meier Curve of Progression-Free Survival (Study 8: ECHELON-3)
Find ADCETRIS® medical information:

Find ADCETRIS® medical information:
ADCETRIS® Quick Finder
Health Professional Information
Clinical Studies
14 CLINICAL STUDIES
14.1 Classical Hodgkin Lymphoma
Randomized Clinical Trial in Previously Untreated Stage III or IV Classical Hodgkin Lymphoma (Study 5: ECHELON-1, NCT01712490)
The efficacy of ADCETRIS in combination with chemotherapy for the treatment of patients with previously untreated Stage III or IV cHL was evaluated in a randomized, open-label, 2-arm, multicenter trial. Of the 1334 total patients, 664 patients were randomized to the ADCETRIS + doxorubicin [A], vinblastine [V] and dacarbazine [D] (ADCETRIS + AVD) arm and 670 patients were randomized to the A+ bleomycin [B] + V + D (ABVD) arm. Patients in both treatment arms were treated intravenously on Days 1 and 15 of each 28-day cycle for up to 6 cycles. Dosing in each treatment arm was administered according to the following:
- •
- ADCETRIS + AVD arm: ADCETRIS 1.2 mg/kg over 30 minutes, doxorubicin 25 mg/m2, vinblastine 6 mg/m2, and dacarbazine 375 mg/m2
- •
- ABVD arm: doxorubicin 25 mg/m2, bleomycin 10 units/m2, vinblastine 6 mg/m2, and dacarbazine 375 mg/m2
Efficacy was established based on modified progression-free survival (modified PFS) per independent review facility (IRF). A modified PFS event is defined as progression, death, or receipt of additional anticancer therapy for patients who are not in a complete response (CR) after completion of frontline therapy.
Patients had Stage III (36%) or IV disease (64%), and 62% had extranodal involvement at diagnosis. Most patients were male (58%) and white (84%). The median age was 36 years (range, 18‑83); 186 patients (14%) were 60 years or older.
The efficacy results are summarized in Table 13 and Figure 1.
| ||
Modified Progression-Free Survival per IRF* | ADCETRIS + AVD | ABVD |
Number of events (%) | 117 (18%) | 146 (22%) |
Median months (95% CI) | NE† | NE† |
Hazard ratio (95% CI)‡ | 0.77 (0.60, 0.98) | |
P-value§ | 0.035 | |
Reason leading to a modified PFS event | ||
Progressive disease | 90 (14) | 102 (15) |
Death due to any cause | 18 (3) | 22 (3) |
Receipt of additional anticancer therapy for | 9 (1) | 22 (3) |
At the time of the modified PFS analysis, the prespecified interim OS analysis did not demonstrate a significant difference. The CR rate per IRF assessment at the end of the randomized regimen was 73% on the ADCETRIS + AVD arm and 70% on the ABVD arm.
A prespecified second interim analysis showed a statistically significant improvement in OS in the ADCETRIS + AVD arm (39 deaths) compared to the ABVD arm (64 deaths). With an estimated median follow-up of 6.1 years, the stratified hazard ratio was 0.59 (95% CI, 0.396; 0.879), with a 2-sided p-value of 0.009 (significance level, 0.0365). Median OS was not reached in either treatment arm (Figure 2).
Randomized Clinical Trial in Previously Untreated High Risk Classical Hodgkin Lymphoma (Study 7, AHOD1331, NCT02166463)
The efficacy of ADCETRIS in combination with chemotherapy for the treatment of pediatric patients (2 to <22 years of age) with previously untreated, high risk cHL was evaluated in a randomized, open-label, actively controlled trial. High risk was defined as Ann Arbor Stage IIB with bulk disease, Stage IIIB, Stage IVA, and Stage IVB. Of the 600 total patients randomized, 300 were randomized to ADCETRIS + Doxorubicin [A], Vincristine [V], Etoposide [E], Prednisone [P], Cyclophosphamide [C] (ADCETRIS + AVEPC) arm and 300 patients were randomized to A+ Bleomycin [B]+V+E+P+C (ABVE-PC) arm. Patients in each treatment arm received up to 5 cycles of the following:
- •
- ADCETRIS + AVEPC arm: ADCETRIS 1.8 mg/kg over 30 minutes (day 1), doxorubicin 25 mg/m2 (days 1 and 2), vincristine 1.4 mg/m2 (day 8), etoposide 125 mg/m2 (days 1-3), prednisone 20 mg/m2 BID (days 1-7), cyclophosphamide 600 mg/m2 (days 1 and 2)
- •
- ABVE-PC arm: doxorubicin 25 mg/m2 (days 1 and 2), bleomycin 5 units/m2 (day1) and 10 units/m2 (day 8), vincristine 1.4 mg/m2 (days 1 and 8), etoposide 125 mg/m2 (days 1-3), prednisone 20 mg/m2 BID (days 1-7), cyclophosphamide 600 mg/m2 (days 1 and 2)
The median age was 15 years (range: 3-21 years); 53% were male, 74% were White, 11% Black, and 3% Asian. Nine patients were <6 years, 81 patients were 6 to <12 years, 448 patients were 12 to <18 years, and 62 patients were ≥18 years. Of the 600 enrolled patients, 20% had disease stage of IIB with bulk disease, 19% had IIIB, 29% had IVA, and 31% had IVB.
Efficacy was established based on event-free-survival (EFS), defined as the time from randomization to the earliest of disease progression or relapse, second malignancy, or death due to any cause. Efficacy results are summarized in Table 14.
| NR = Not reached. | ||
| ||
Event-Free Survival | ADCETRIS + AVEPC | ABVE-PC |
Number of Events (%) | 23 (8) | 52 (17) |
Median (95% CI) | NR | NR |
Hazard Ratio (95% CI)* | 0.41 (0.25, 0.67) | |
P-value (log-rank test)† | 0.0002 | |
Randomized Placebo-Controlled Clinical Trial in Classical Hodgkin Lymphoma Post-Auto-HSCT Consolidation (Study 3: AETHERA, NCT01100502)
The efficacy of ADCETRIS in patients with cHL at high risk of relapse or disease progression post-auto-HSCT was studied in a randomized, double-blind, placebo-controlled clinical trial. Three hundred twenty-nine (329) patients were randomized 1:1 to receive placebo or ADCETRIS 1.8 mg/kg intravenously over 30 minutes every 3 weeks for up to 16 cycles, beginning 30–45 days post-auto-HSCT. Patients in the placebo arm with progressive disease per investigator could receive ADCETRIS as part of a separate trial. The primary endpoint was progression-free survival (PFS) determined by independent review facility (IRF). Standard international guidelines were followed for infection prophylaxis for HSV, VZV, and PJP post-auto-HSCT [see Clinical Trial Experience (6.1)].
High risk of post-auto-HSCT relapse or progression was defined according to status following frontline therapy: refractory, relapse within 12 months, or relapse ≥12 months with extranodal disease. Patients were required to have obtained a complete response (CR), partial response (PR), or stable disease (SD) to most recent pre-auto-HSCT salvage therapy.
A total of 329 patients were enrolled and randomized (165 ADCETRIS, 164 placebo); 327 patients received study treatment. Patient demographics and baseline characteristics were generally balanced between treatment arms. The 329 patients ranged in age from 18–76 years (median, 32 years) and most were male (53%) and white (94%). Patients had received a median of 2 prior systemic therapies (range, 2–8) excluding autologous hematopoietic stem cell transplantation.
The efficacy results are summarized in Table 15. PFS is calculated from randomization to date of disease progression or death (due to any cause). The median PFS follow-up time from randomization was 22 months (range, 0–49). Study 3 (AETHERA) demonstrated a statistically significant improvement in IRF-assessed PFS and increase in median PFS in the ADCETRIS arm compared with the placebo arm. At the time of the PFS analysis, an interim overall survival analysis demonstrated no difference.
Progression-Free Survival per IRF | ADCETRIS | Placebo |
Number of events (%) | 60 (36) | 75 (46) |
Median months (95% CI) | 24.1 (11.5, NE†) | |
Stratified Hazard Ratio (95% CI) | 0.57 (0.40, 0.81) | |
Stratified Log-Rank Test P-value | 0.001 | |
Clinical Trial in Relapsed Classical Hodgkin Lymphoma (Study 1, NCT00848926)
The efficacy of ADCETRIS in patients with cHL who relapsed after autologous hematopoietic stem cell transplantation was evaluated in one open-label, single-arm, multicenter trial. One hundred two (102) patients were treated with 1.8 mg/kg of ADCETRIS intravenously over 30 minutes every 3 weeks. An independent review facility (IRF) performed efficacy evaluations which included overall response rate (ORR = complete response [CR] + partial response [PR]) and duration of response as defined by clinical and radiographic measures including computed tomography (CT) and positron-emission tomography (PET) as defined in the 2007 Revised Response Criteria for Malignant Lymphoma (modified).
The 102 patients ranged in age from 15–77 years (median, 31 years) and most were female (53%) and white (87%). Patients had received a median of 5 prior therapies including autologous hematopoietic stem cell transplantation.
The efficacy results are summarized in Table 16. Duration of response is calculated from date of first response to date of progression or data cutoff date.
14.2 Systemic Anaplastic Large Cell Lymphoma and Other CD30-Expressing Peripheral T-Cell Lymphomas
Randomized Clinical Trial in Previously Untreated Systemic Anaplastic Large Cell Lymphoma or Other CD30-Expressing Peripheral T-Cell Lymphomas (Study 6: ECHELON-2, NCT01777152)
The efficacy of ADCETRIS in combination with chemotherapy for the treatment of adult patients with previously untreated, CD30-expressing PTCL was evaluated in a multicenter, randomized, double-blind, double-dummy, actively controlled trial. For enrollment, the trial required CD30 expression ≥10% per immunohistochemistry. The trial excluded patients with primary cutaneous CD30-positive T-cell lymphoproliferative disorders and lymphomas. The trial required hepatic transaminases ≤3 times ULN, total bilirubin ≤1.5 times ULN, and serum creatinine ≤2 times ULN.
Of the 452 total patients, 226 patients were randomized to the ADCETRIS + CHP arm and 226 patients were randomized to the CHOP arm. Patients in both treatment arms were treated intravenously on Day 1 of each 21-day cycle for 6 to 8 cycles; prednisone was administered orally on Days 1‑5. Dosing in each treatment arm was administered according to the following:
- •
- ADCETRIS + CHP arm: ADCETRIS 1.8 mg/kg over 30 minutes, cyclophosphamide 750 mg/m2, doxorubicin 50 mg/m2, and prednisone 100 mg orally
- •
- CHOP arm: cyclophosphamide 750 mg/m2, doxorubicin 50 mg/m2, vincristine 1.4 mg/m2, and prednisone 100 mg orally
The median age was 58 years (range: 18 to 85), 63% were male, 62% were White, 22% were Asian, and 78% had an ECOG performance status of 0-1. Of the 452 patients enrolled, the disease subtypes included patients with systemic ALCL [70%; 48% anaplastic lymphoma kinase (ALK) negative and 22% ALK positive], PTCL NOS (16%), angioimmunoblastic T-cell lymphoma (12%), adult T-cell leukemia/lymphoma (2%), and enteropathy-associated T-cell lymphoma (<1%). Most patients had Stage III or IV disease (81%) and a baseline international prognostic index of 2 or 3 (63%).
During randomized treatment, on the ADCETRIS + CHP arm, 70% of patients received 6 cycles and 18% of patients received 8 cycles. On the CHOP arm, 62% of patients received 6 cycles and 19% received 8 cycles.
Efficacy was based on IRF-assessed PFS, which was defined as time from randomization to progression, death due to any cause, or receipt of subsequent anticancer chemotherapy to treat residual or progressive disease. Other efficacy endpoints included PFS in patients with systemic ALCL, overall survival, complete response rate, and overall response rate. Efficacy results are summarized in Table 17. Kaplan-Meier curves for PFS and overall survival are presented in Figure 5 and Figure 6, respectively.
| NE = Not estimable | ||
| ||
Outcomes per IRF* | ADCETRIS + CHP | CHOP |
PFS | ||
Number of events, n (%) | 95 (42) | 124 (55) |
Median PFS, months (95% CI) | 48.2 (35.2, NE) | 20.8 (12.7, 47.6) |
Hazard ratio (95% CI)† | 0.71 (0.54, 0.93) | |
P-value‡ | 0.011 | |
Reason leading to a PFS event, n (%) | ||
Progressive disease | 71 (31) | 86 (38) |
Death | 13 (6) | 17 (8) |
Receipt of subsequent anticancer chemotherapy to treat residual or progressive disease | 11 (5) | 21 (9) |
PFS for patients with sALCL | ||
N | 163 | 151 |
Number of patients with a PFS event, n (%) | 56 (34) | 73 (48) |
Median PFS, months (95% CI) | 55.7 (48.2, NE) | 54.2 (13.4, NE) |
Hazard ratio (95% CI)† | 0.59 (0.42, 0.84) | |
P-value‡ | 0.003 | |
OS§ | ||
Number of deaths | 51 (23) | 73 (32) |
Median OS, months (95% CI) | NE (NE, NE) | NE (54.2, NE) |
Hazard ratio (95% CI)† | 0.66 (0.46, 0.95) | |
P-value‡ | 0.024 | |
CR Rate¶ | ||
% (95% CI) | 68 (61, 74) | 56 (49, 62) |
P-value# | 0.007 | |
ORR¶ | ||
% (95% CI) | 83 (78, 88) | 72 (66, 78) |
P-value# | 0.003 | |
14.3 Systemic Anaplastic Large Cell Lymphoma
Clinical Trial in Relapsed sALCL (Study 2, NCT00866047)
The efficacy of ADCETRIS in patients with relapsed sALCL was evaluated in one open-label, single-arm, multicenter trial. This trial included patients who had sALCL that was relapsed after prior therapy. Fifty-eight (58) patients were treated with 1.8 mg/kg of ADCETRIS administered intravenously over 30 minutes every 3 weeks. An IRF performed efficacy evaluations which included overall response rate (ORR = complete response [CR] + partial response [PR]) and duration of response as defined by clinical and radiographic measures including computed tomography (CT) and positron-emission tomography (PET) as defined in the 2007 Revised Response Criteria for Malignant Lymphoma (modified).
The 58 patients ranged in age from 14–76 years (median, 52 years) and most were male (57%) and white (83%). Patients had received a median of 2 prior therapies; 26% of patients had received prior autologous hematopoietic stem cell transplantation. Fifty percent (50%) of patients were relapsed, and 50% of patients were refractory to their most recent prior therapy. Seventy-two percent (72%) were anaplastic lymphoma kinase (ALK)-negative.
The efficacy results are summarized in Table 18. Duration of response is calculated from date of first response to date of progression or data cutoff date.
14.4 Primary Cutaneous Anaplastic Large Cell Lymphoma and CD30-Expressing Mycosis Fungoides
Randomized Clinical Trial in Primary Cutaneous Anaplastic Large Cell Lymphoma and CD30-expressing Mycosis Fungoides (Study 4: ALCANZA, NCT01578499)
The efficacy of ADCETRIS in patients with primary cutaneous anaplastic large cell lymphoma (pcALCL) or mycosis fungoides (MF) requiring systemic therapy was studied in ALCANZA, a randomized, open-label, multicenter clinical trial. In ALCANZA, one hundred thirty-one (131) patients were randomized 1:1 to receive ADCETRIS 1.8 mg/kg intravenously over 30 minutes every 3 weeks or physician’s choice of methotrexate (5 to 50 mg orally weekly) or bexarotene (300 mg/m2 orally daily). The randomization was stratified by baseline disease diagnosis (MF or pcALCL). Patients could receive a maximum of 16 cycles (21-day cycle) of therapy every 3 weeks for those receiving brentuximab vedotin or 48 weeks of therapy for those in the control arm.
Patients with pcALCL must have received prior radiation or systemic therapy, and must have at least 1 biopsy with CD30-expression of ≥10%. Patients with MF must have received prior systemic therapy and have had skin biopsies from at least 2 separate lesions, with CD30-expression of ≥10% in at least 1 biopsy.
A total of 131 patients were randomized (66 ADCETRIS, 65 physician’s choice). The efficacy results were based on 128 patients (64 patients in each arm with CD30-expression of ≥10% in at least one biopsy). Among 128 patients, the patients’ age ranged from 22–83 years (median, 60 years), and 55% of them were male and 85% of them were white. Patients had received a median of 4 prior therapies (range, 0–15), including a median of 1 prior skin-directed therapy (range, 0–9) and 2 systemic therapies (range, 0–11). At study entry, patients were diagnosed as Stage 1 (25%), Stage 2 (38%), Stage 3 (5%), or Stage 4 (13%).
Efficacy was established based on the proportion of patients achieving an objective response (CR+PR) that lasts at least 4 months (ORR4). ORR4 was determined by independent review facility (IRF) using the global response score (GRS), consisting of skin evaluations per modified severity-weighted assessment tool (mSWAT), nodal and visceral radiographic assessment, and detection of circulating Sézary cells (MF patients only). Additional efficacy outcome measures included proportion of patients achieving a complete response (CR) per IRF, and progression-free survival (PFS) per IRF.
The efficacy results are summarized in Table 19 below and the Kaplan-Meier curves of IRF-assessed PFS are shown in Figure 7.
| ||
ADCETRIS | Physician’s Choice* | |
ORR4† | ||
Percent (95% CI‡) | 56.3 (44.1, 68.4) | 12.5 (4.4, 20.6) |
P-value§ | <0.001 | |
ORR | 67.2 (55.7, 78.7) | 20.3 (10.5, 30.2) |
CR | ||
Percent (95% CI‡) | 15.6 (7.8, 26.9) | 1.6 (0, 8.4) |
0.0066 | ||
PR | 51.6 (39.3, 63.8) | 18.8 (9.2, 28.3) |
PFS | ||
Number of events (%) | 36 (56.3) | 50 (78.1) |
Median months (95% CI‡) | 16.7 (14.9, 22.8) | 3.5 (2.4, 4.6) |
Hazard Ratio (95% CI‡) | 0.27 (0.17, 0.43) | |
<0.001 | ||
Supportive trials include 2 single-arm trials, which enrolled patients with MF who were treated with ADCETRIS 1.8 mg/kg intravenously over 30 minutes every 3 weeks. Out of 73 patients with MF from the 2 pooled supportive trials, 34% (25/73) achieved ORR4. Among these 73 patients, 35 had 1% to 9% CD30-expression and 31% (11/35) achieved ORR4.
14.5 Relapsed or Refractory Large B-Cell Lymphoma (LBCL)
Randomized Clinical Trial in Relapsed or Refractory Large B-Cell Lymphoma (Study 8: ECHELON-3)
The efficacy of ADCETRIS in combination with lenalidomide and a rituximab product for the treatment of adult patients with relapsed or refractory LBCL after two or more lines of systemic therapy was evaluated in ECHELON-3 (NCT04404283), a randomized, multicenter, double-blind, placebo-controlled trial. Eligible patients were 18 years of age and older with relapsed or refractory LBCL, including DLBCL NOS, DLBCL arising from indolent lymphoma, or HGBL, an ECOG performance status of 0–2, and were not eligible to receive an auto-HSCT or CAR T-cell therapy. The study excluded patients with active CNS lymphoma or Grade 2 or higher peripheral neuropathy.
Patients were randomized 1:1 to receive ADCETRIS plus lenalidomide and a rituximab product or to receive placebo plus lenalidomide and a rituximab product until disease progression or unacceptable toxicity. Randomization was stratified by CD30 expression (≥1% versus <1%), prior allogenic or autologous HSCT therapy, prior CAR T-cell therapy and cell of origin (germinal-center B-cell like [GCB] or non-GCB). Dosing in each treatment arms as follows:
- •
- ADCETRIS plus lenalidomide and rituximab arm: ADCETRIS 1.2 mg/kg via intravenous infusion every 3 weeks, lenalidomide 20 mg orally daily, and rituximab 375 mg/m2 via intravenous infusion every 3 weeks. Starting with Cycle 2, rituximab intravenous treatment could be substituted with rituximab 1,400 mg and hyaluronidase human 23,400 Units via subcutaneous injection every 3 weeks.
- •
- Placebo plus lenalidomide and rituximab arm: lenalidomide 20 mg orally daily, and rituximab 375 mg/m2 via intravenous infusion every 3 weeks. Starting with Cycle 2, rituximab intravenous treatment could be substituted with rituximab 1,400 mg and hyaluronidase human 23,400 Units via subcutaneous injection every 3 weeks.
Prophylaxis with granulocyte-colony stimulating factor (G-CSF) was mandated for both arms.
Of the 230 patients randomized (112 to ADCETRIS plus lenalidomide and rituximab, 118 to placebo plus lenalidomide and rituximab), the median age was 71 years (range 21 to 89 years); 57% were male, 53% were White, 26% were Asian and 4% were Hispanic or Latino. Race and ethnicity was not reported in 46 (20%) patients. There were no Black or African American patients enrolled. Of the total 230 patients, 71% had DLBCL NOS, 26% had transformed disease from prior indolent lymphoma, 16% had HGBL with MYC and BCL2 and/or BCL6 rearrangements or HGBL NOS. Of the patients with DLBCL NOS, 73 (45%) and 91 (55%) had GCB and non-GCB subtypes, respectively. Twenty-nine percent of patients had received prior CAR T-cell therapy and 12% had received prior HSCT. The median number of prior systemic therapies was 3 (range 2-8) with 41% receiving 2 prior therapies and 50% receiving 3 or more prior therapies. Eighty four percent had refractory disease to last therapy.
The major efficacy outcome was overall survival (OS). Additional efficacy outcome measures included progression free survival (PFS) and Objective Response Rate (ORR) according to investigator assessment per the Lugano Criteria for Response Assessment 2014. The results are presented in Table 20 and Figures 8 and 9.
| CI = confidence interval; CR = complete response; HR = hazard ratio; PFS = progression free survival; OS = overall survival. Stratified analyses were based on the stratification factors of cell of origin (GCB, non-GCB) and CD30 status (≥1%, <1%) at randomization. | ||
| ||
ADCETRIS + Lenalidomide + Rituximab (N=112) | Placebo + Lenalidomide + Rituximab (N=118) | |
Overall Survival | ||
Deaths, n (%) | 58 (51.8) | 76 (64.4) |
Median, months (95% CI) | 13.8 (10.3, 18.8) | 8.5 (5.4, 11.7) |
HR (95% CI)* | 0.63 (0.45, 0.89) | |
0.0085 | ||
PFS per Investigator | ||
Number (%) of patients with event | 71 (63.4) | 85 (72.0) |
Progressive disease, n (%) | 56 (50.0) | 71 (60.2) |
Death, n (%) | 15 (13.4) | 14 (11.9) |
Median, months (95% CI) | 4.2 (2.9, 7.1) | 2.6 (1.4, 3.1) |
HR (95% CI)* | 0.53 (0.38, 0.73) | |
p-value† | <0.0001 | |
Objective Response Rate per Investigator | ||
Objective response rate (CR or PR), n (%) | 72 (64.3) | 49 (41.5) |
95% CI for ORR§ | (54.7, 73.1) | (32.5, 51.0) |
p-value¶ | 0.0006 | |
Complete response (CR), n (%) | 45 (40.2) | 22 (18.6) |
Figure 8: Kaplan-Meier Curve of Overall Survival (Study 8: ECHELON-3)
Figure 9: Kaplan-Meier Curve of Progression-Free Survival (Study 8: ECHELON-3)
Health Professional Information
{{section_name_patient}}
{{section_body_html_patient}}
Resources
Didn’t find what you were looking for? Contact us.
Chat online with Pfizer Medical Information regarding your inquiry on a Pfizer medicine.
*Speak with a Pfizer Medical Information Professional regarding your medical inquiry. Available 9AM-5PM ET Monday to Friday; excluding holidays.
Submit a medical question for Pfizer prescription products.
Report Adverse Event
Pfizer Safety
To report an adverse event related to the Pfizer-BioNTech COVID-19 Vaccine, and you are not part of a clinical trial* for this product, click the link below to submit your information:
Pfizer Safety Reporting Site*If you are involved in a clinical trial for this product, adverse events should be reported to your coordinating study site.
If you cannot use the above website, or would like to report an adverse event related to a different Pfizer product, please call Pfizer Safety at (800) 438-1985.
FDA Medwatch
You may also contact the U.S. Food and Drug Administration (FDA) directly to report adverse events or product quality concerns either online at www.fda.gov/medwatch or call (800) 822-7967.