CELONTIN®
(methsuximide)
Find CELONTIN® medical information:
Find CELONTIN® medical information:
CELONTIN® Quick Finder
Indications and Usage
Dosage and Administration
DOSAGE AND ADMINISTRATION
Optimum dosage of Celontin must be determined by trial. A suggested dosage schedule is 300 mg per day for the first week. If required, dosage may be increased thereafter at weekly intervals by 300 mg per day for the three weeks following to a daily dosage of 1.2 g. Because therapeutic effect and tolerance vary among patients, therapy with Celontin must be individualized according to the response of each patient. Optimal dosage is that amount of Celontin which is barely sufficient to control seizures so that side effects may be kept to a minimum.
Celontin may be administered in combination with other anticonvulsants when other forms of epilepsy coexist with absence (petit mal).
Contraindications
Warnings and Precautions
WARNINGS
Blood dyscrasias
Blood dyscrasias, including some with fatal outcome, have been reported to be associated with the use of succinimides; therefore, periodic blood counts should be performed. Should signs and/or symptoms of infection (eg, sore throat, fever) develop, blood counts should be considered at that point.
Effects on Liver
It has been reported that succinimides have produced morphological and functional changes in animal liver. For this reason, methsuximide should be administered with extreme caution to patients with known liver or renal disease. Periodic urinalysis and liver function studies are advised for all patients receiving the drug.
Systemic Lupus Erythematosus
Cases of systemic lupus erythematosus have been reported with the use of succinimides. The physician should be alert to this possibility.
Suicidal Behavior and Ideation
Antiepileptic drugs (AEDs), including Celontin, increase the risk of suicidal thoughts or behavior in patients taking these drugs for any indication. Patients treated with any AED for any indication should be monitored for the emergence or worsening of depression, suicidal thoughts or behavior, and/or any unusual changes in mood or behavior.
Pooled analyses of 199 placebo-controlled clinical trials (mono- and adjunctive therapy) of 11 different AEDs showed that patients randomized to one of the AEDs had approximately twice the risk (adjusted Relative Risk 1.8, 95% CI:1.2, 2.7) of suicidal thinking or behavior compared to patients randomized to placebo. In these trials, which had a median treatment duration of 12 weeks, the estimated incidence rate of suicidal behavior or ideation among 27,863 AED-treated patients was 0.43%, compared to 0.24% among 16,029 placebo-treated patients, representing an increase of approximately one case of suicidal thinking or behavior for every 530 patients treated. There were four suicides in drug-treated patients in the trials and none in placebo-treated patients, but the number is too small to allow any conclusion about drug effect on suicide.
The increased risk of suicidal thoughts or behavior with AEDs was observed as early as one week after starting drug treatment with AEDs and persisted for the duration of treatment assessed. Because most trials included in the analysis did not extend beyond 24 weeks, the risk of suicidal thoughts or behavior beyond 24 weeks could not be assessed.
The risk of suicidal thoughts or behavior was generally consistent among drugs in the data analyzed. The finding of increased risk with AEDs of varying mechanisms of action and across a range of indications suggests that the risk applies to all AEDs used for any indication. The risk did not vary substantially by age (5–100 years) in the clinical trials analyzed.
Table 1 shows absolute and relative risk by indication for all evaluated AEDs.
| Indication | Placebo Patients with Events Per 1000 Patients | Drug Patients with Events Per 1000 Patients | Relative Risk: Incidence of Events in Drug Patients/Incidence in Placebo Patients | Risk Difference: Additional Drug Patients with Events Per 1000 Patients |
|---|---|---|---|---|
Epilepsy | 1.0 | 3.4 | 3.5 | 2.4 |
Psychiatric | 5.7 | 8.5 | 1.5 | 2.9 |
Other | 1.0 | 1.8 | 1.9 | 0.9 |
Total | 2.4 | 4.3 | 1.8 | 1.9 |
The relative risk for suicidal thoughts or behavior was higher in clinical trials for epilepsy than in clinical trials for psychiatric or other conditions, but the absolute risk differences were similar for the epilepsy and psychiatric indications.
Anyone considering prescribing Celontin or any other AED must balance the risk of suicidal thoughts or behavior with the risk of untreated illness. Epilepsy and many other illnesses for which AEDs are prescribed are themselves associated with morbidity and mortality and an increased risk of suicidal thoughts and behavior. Should suicidal thoughts and behavior emerge during treatment, the prescriber needs to consider whether the emergence of these symptoms in any given patient may be related to the illness being treated.
Patients, their caregivers, and families should be informed that AEDs increase the risk of suicidal thoughts and behavior and should be advised of the need to be alert for the emergence or worsening of the signs and symptoms of depression, any unusual changes in mood or behavior, or the emergence of suicidal thoughts, behavior, or thoughts about self-harm. Behaviors of concern should be reported immediately to healthcare providers.
Usage in Pregnancy
Reports suggest an association between the use of anticonvulsant drugs by women with epilepsy and an elevated incidence of birth defects in children born to these women. Data are more extensive with respect to phenytoin and phenobarbital, but these are also the most commonly prescribed anticonvulsants; less systematic or anecdotal reports suggest a possible similar association with the use of all known anticonvulsant drugs.
The reports suggesting an elevated incidence of birth defects in children of drug-treated epileptic women cannot be regarded as adequate to prove a definite cause and effect relationship. There are intrinsic methodologic problems in obtaining adequate data on drug teratogenicity in humans; the possibility also exists that other factors, eg, genetic factors or the epileptic condition itself, may be more important than drug therapy in leading to birth defects. The great majority of mothers on anticonvulsant medication deliver normal infants. It is important to note that anticonvulsant drugs should not be discontinued in patients in whom the drug is administered to prevent major seizures because of the strong possibility of precipitating status epilepticus with attendant hypoxia and threat to life. In individual cases where the severity and frequency of the seizure disorder are such that the removal of medication does not pose a serious threat to the patient, discontinuation of the drug may be considered prior to and during pregnancy, although it cannot be said with any confidence that even minor seizures do not pose some hazard to the developing embryo or fetus.
The prescribing physician will wish to weigh these considerations in treating or counseling epileptic women of childbearing potential.
PRECAUTIONS
General
It is recommended that the physician withdraw the drug slowly on the appearance of unusual depression, aggressiveness, or other behavioral alterations.
As with other anticonvulsants, it is important to proceed slowly when increasing or decreasing dosage, as well as when adding or eliminating other medication. Abrupt withdrawal of anticonvulsant medication may precipitate absence (petit mal) status.
Methsuximide, when used alone in mixed types of epilepsy, may increase the frequency of grand mal seizures in some patients.
Information for Patients
Inform patients of the availability of a Medication Guide, and instruct them to read the Medication Guide prior to taking Celontin. Instruct patients to take Celontin only as prescribed.
Methsuximide may impair the mental and/or physical abilities required for the performance of potentially hazardous tasks, such as driving a motor vehicle or other such activity requiring alertness; therefore, the patient should be cautioned accordingly. Patients taking methsuximide should be advised of the importance of adhering strictly to the prescribed dosage regimen.
Patients should be instructed to promptly contact their physician if they develop signs and/or symptoms suggesting an infection (eg, sore throat, fever).
ADVICE TO THE PHARMACIST AND PATIENT: Since methsuximide has a relatively low melting temperature (124° F), storage conditions which may promote high temperatures (closed cars, delivery vans, or storage near steam pipes) should be avoided. Do not dispense or use capsules that are not full or in which contents have melted. Effectiveness may be reduced. Protect from excessive heat (104° F).
Patients, their caregivers, and families should be counseled that AEDs, including Celontin, may increase the risk of suicidal thoughts and behavior and should be advised of the need to be alert for the emergence or worsening of symptoms of depression, any unusual changes in mood or behavior, or the emergence of suicidal thoughts, behavior, or thoughts about self-harm. Behaviors of concern should be reported immediately to healthcare providers.
Patients should be encouraged to enroll in the North American Antiepileptic Drug (NAAED) Pregnancy Registry if they become pregnant. This registry is collecting information about the safety of antiepileptic drugs during pregnancy. To enroll, patients can call the toll free number 1-888-233-2334 (see PRECAUTIONS: Pregnancy section).
Drug Interactions
Since Celontin (methsuximide) may interact with concurrently administered antiepileptic drugs, periodic serum level determinations of these drugs may be necessary (eg, methsuximide may increase the plasma concentrations of phenytoin and phenobarbital).
Pregnancy
To provide information regarding the effects of in utero exposure to Celontin, physicians are advised to recommend that pregnant patients taking Celontin enroll in the (NAAED) Pregnancy Registry. This can be done by calling the toll free number 1‑888‑233‑2334, and must be done by patients themselves. Information on the registry can also be found at the website:
https://www.aedpregnancyregistry.org/.
See WARNINGS.
Adverse Reactions
ADVERSE REACTIONS
Gastrointestinal System: Gastrointestinal symptoms occur frequently and have included nausea or vomiting, anorexia, diarrhea, weight loss, epigastric and abdominal pain, and constipation.
Hemopoietic System: Hemopoietic complications associated with the administration of methsuximide have included eosinophilia, leukopenia, monocytosis, and pancytopenia with or without bone marrow suppression.
Nervous System: Neurologic and sensory reactions reported during therapy with methsuximide have included drowsiness, ataxia or dizziness, irritability and nervousness, headache, blurred vision, photophobia, hiccups, and insomnia. Drowsiness, ataxia, and dizziness have been the most frequent side effects noted. Psychologic abnormalities have included confusion, instability, mental slowness, depression, hypochondriacal behavior, and aggressiveness. There have been rare reports of psychosis, suicidal behavior, and auditory hallucinations.
Integumentary System: Dermatologic manifestations which have occurred with the administration of methsuximide have included urticaria, Stevens-Johnson syndrome, and pruritic erythematous rashes.
Cardiovascular: Hyperemia.
Genitourinary System: Proteinuria, microscopic hematuria.
Body as a Whole: Periorbital edema.
Overdosage
OVERDOSAGE
Acute overdoses may produce nausea, vomiting, and CNS depression including coma with respiratory depression. Methsuximide poisoning may follow a biphasic course. Following an initial comatose state, patients have awakened and then relapsed into a coma within 24 hours. It is believed that an active metabolite of methsuximide, N-desmethylmethsuximide, is responsible for this biphasic profile. It is important to follow plasma levels of N-desmethylmethsuximide in methsuximide poisonings. Levels greater than 40 µg/mL have caused toxicity, and coma has been seen at levels of 150 µg/mL.
Treatment
Treatment should include emesis (unless the patient is or could rapidly become obtunded, comatose, or convulsing) or gastric lavage, activated charcoal, cathartics, and general supportive measures. Charcoal hemoperfusion may be useful in removing the N-desmethyl metabolite of methsuximide. Forced diuresis and exchange transfusions are ineffective.
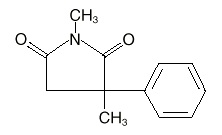
Description
DESCRIPTION
Celontin (methsuximide) is an anticonvulsant succinimide, chemically designated as N,2-Dimethyl-2-phenylsuccinimide, with the following structural formula:
Each Celontin capsule contains 300 mg methsuximide, USP. Also contains starch, NF. The capsule contains colloidal silicon dioxide, NF; D&C yellow No. 10; FD&C yellow No. 6; gelatin, NF; and sodium lauryl sulfate, NF.
Clinical Pharmacology
CLINICAL PHARMACOLOGY
Methsuximide suppresses the paroxysmal three cycle per second spike and wave activity associated with lapses of consciousness which is common in absence (petit mal) seizures. The frequency of epileptiform attacks is reduced, apparently by depression of the motor cortex and elevation of the threshold of the central nervous system to convulsive stimuli.
How Supplied/Storage and Handling
Medication Guide
MEDICATION GUIDE
CELONTIN (Suh lŏn' tĭn)
(methsuximide)
Capsules
Read this Medication Guide before you start taking CELONTIN and each time you get a refill. There may be new information. This information does not take the place of talking to your healthcare provider about your medical condition or treatment. If you have any questions about CELONTIN, ask your healthcare provider or pharmacist.
What is the most important information I should know about CELONTIN?
Do not stop taking CELONTIN without first talking to your healthcare provider. Stopping CELONTIN suddenly can cause serious problems.
CELONTIN can cause serious side effects, including:
- 1.
- Rare but serious blood problems that may be life-threatening. Call your healthcare provider right away if you have:
- o
- fever, swollen glands, or sore throat that come and go or do not go away
- o
- frequent infections or an infection that does not go away
- o
- easy bruising
- o
- red or purple spots on your body
- o
- bleeding gums or nose bleeds
- o
- severe fatigue or weakness
- 2.
- Systematic Lupus Erythematosus. Call your healthcare provider right away if you have any of these symptoms:
- o
- joint pain and swelling
- o
- muscle pain
- o
- fatigue
- o
- low-grade fever
- o
- pain in the chest that is worse with breathing
- o
- unexplained skin rash
- 3.
- Like other antiepileptic drugs, CELONTIN may cause suicidal thoughts or actions in a very small number of people, about 1 in 500.
Call a healthcare provider right away if you have any of these symptoms, especially if they are new, worse, or worry you:
- o
- thoughts about suicide or dying
- o
- attempts to commit suicide
- o
- new or worse depression
- o
- new or worse anxiety
- o
- feeling agitated or restless
- o
- panic attacks
- o
- trouble sleeping (insomnia)
- o
- new or worse irritability
- o
- acting aggressive, being angry, or violent
- o
- acting on dangerous impulses
- o
- an extreme increase in activity and talking (mania)
- o
- other unusual changes in behavior or mood
- How can I watch for early symptoms of suicidal thoughts and actions?
- o
- Pay attention to any changes, especially sudden changes, in mood, behaviors, thoughts, or feelings.
- o
- Keep all follow-up visits with your healthcare provider as scheduled.
- Call your healthcare provider between visits as needed, especially if you are worried about symptoms.
- Do not stop CELONTIN without first talking to a healthcare provider.
- Stopping CELONTIN suddenly can cause serious problems. Stopping a seizure medicine suddenly in a patient who has epilepsy can cause seizures that will not stop (status epilepticus).
- Suicidal thoughts or actions can be caused by things other than medicines. If you have suicidal thoughts or actions, your healthcare provider may check for other causes.
What is CELONTIN?
CELONTIN is a prescription medicine used to treat absence (petit mal) seizures that have not gotten better with other seizure medicines.
Who should not take CELONTIN?
Do not take CELONTIN if you are allergic to succinimides (methsuximide or ethosuximide) or any of the ingredients in CELONTIN. See the end of this Medication Guide for a complete list of ingredients in CELONTIN.
What should I tell my healthcare provider before taking CELONTIN?
Before you take CELONTIN, tell your healthcare provider if you:
- •
- have or have had liver problems
- •
- have or have had depression, mood problems or suicidal thoughts or behavior
- •
- have any other medical conditions
- •
- are pregnant or plan to become pregnant. It is not known if CELONTIN can harm your unborn baby. Tell your healthcare provider right away if you become pregnant while taking CELONTIN. You and your healthcare provider will decide if you should take CELONTIN while you are pregnant.
- o
- If you become pregnant while taking CELONTIN, talk to your healthcare provider about registering with the North American Antiepileptic Drug (NAAED) Pregnancy Registry. The purpose of this registry is to collect information about the safety of antiepileptic drugs during pregnancy. You can enroll in this registry by calling 1‑888‑233‑2334.
- •
- are breastfeeding or plan to breastfeed. It is not known if CELONTIN can pass into breast milk. You and your healthcare provider should decide how you will feed your baby while you take CELONTIN.
Tell your healthcare provider about all the medicines you take, including prescription and non-prescription medicines, vitamins, and herbal supplements. Taking CELONTIN with certain other medicines can cause side effects or affect how well they work. Do not start or stop other medicines without talking to your healthcare provider.
Know the medicines you take. Keep a list of them with you to show your healthcare provider and pharmacist each time you get a new medicine.
How should I take CELONTIN?
- •
- Take CELONTIN exactly as prescribed. Your healthcare provider will tell you how much CELONTIN to take.
- •
- Your healthcare provider may change your dose. Do not change your dose of CELONTIN without talking to your healthcare provider.
- •
- If you take too much CELONTIN, call your healthcare provider or your local Poison Control Center right away.
What should I avoid while taking CELONTIN?
- •
- Do not drink alcohol or take other medicines that make you sleepy or dizzy while taking CELONTIN without first discussing this with your healthcare provider. CELONTIN taken with alcohol or medicines that cause sleepiness or dizziness may make your sleepiness or dizziness worse.
- •
- Do not drive, operate heavy machinery, or do other dangerous activities until you know how CELONTIN affects you. CELONTIN can slow your thinking and motor skills.
What are the possible side effects of CELONTIN?
- •
- See "What is the most important information I should know about CELONTIN?"
CELONTIN may cause other serious side effects, including:
- •
- Grand mal seizures can happen more often or become worse
The most common side effects of CELONTIN include:
|
|
Tell your healthcare provider about any side effect that bothers you or that does not go away.
These are not all the possible side effects with CELONTIN. For more information, ask your healthcare provider or pharmacist.
Call your doctor for medical advice about side effects. You may report side effects to FDA at 1‑800‑FDA‑1088.
How should I store CELONTIN?
- •
- Store CELONTIN at room temperature, between 59°F to 86°F (15°C to 30°C).
- •
- Keep CELONTIN capsules in a dry place.
- •
- Keep CELONTIN out of the light.
- •
- Protect CELONTIN from heat.
- •
- Do not use CELONTIN capsules that if they do not look full or if the contents have melted.
Keep CELONTIN and all medicines out of the reach of children.
General information about CELONTIN
Medicines are sometimes prescribed for purposes other than those listed in a Medication Guide. Do not use CELONTIN for a condition for which it was not prescribed. Do not give CELONTIN to other people, even if they have the same condition. It may harm them.
This Medication Guide summarizes the most important information about CELONTIN. If you would like more information, talk with your healthcare provider. You can ask your healthcare provider or pharmacist for information about CELONTIN that is written for healthcare professionals.
For more information, go to www.pfizer.com or call 1-800-438-1985.
What are the ingredients in CELONTIN?
Active ingredient: methsuximide
Inactive ingredients: starch, colloidal silicon dioxide NF, D&C yellow No. 10, FD&C yellow No.6, gelatin NF, and sodium lauryl sulfate NF.
This Medication Guide has been approved by the U.S. Food and Drug Administration.
LAB-0404-3.0
Revised March 2023
Other
MEDICATION GUIDE
MEDICATION GUIDE
CELONTIN (Suh lŏn' tĭn)
(methsuximide)
Capsules
Read this Medication Guide before you start taking CELONTIN and each time you get a refill. There may be new information. This information does not take the place of talking to your healthcare provider about your medical condition or treatment. If you have any questions about CELONTIN, ask your healthcare provider or pharmacist.
What is the most important information I should know about CELONTIN?
What is the most important information I should know about CELONTIN?
Do not stop taking CELONTIN without first talking to your healthcare provider. Stopping CELONTIN suddenly can cause serious problems.
CELONTIN can cause serious side effects, including:
- 1.
- Rare but serious blood problems that may be life-threatening. Call your healthcare provider right away if you have:
- o
- fever, swollen glands, or sore throat that come and go or do not go away
- o
- frequent infections or an infection that does not go away
- o
- easy bruising
- o
- red or purple spots on your body
- o
- bleeding gums or nose bleeds
- o
- severe fatigue or weakness
- 2.
- Systematic Lupus Erythematosus. Call your healthcare provider right away if you have any of these symptoms:
- o
- joint pain and swelling
- o
- muscle pain
- o
- fatigue
- o
- low-grade fever
- o
- pain in the chest that is worse with breathing
- o
- unexplained skin rash
- 3.
- Like other antiepileptic drugs, CELONTIN may cause suicidal thoughts or actions in a very small number of people, about 1 in 500.
Call a healthcare provider right away if you have any of these symptoms, especially if they are new, worse, or worry you:
- o
- thoughts about suicide or dying
- o
- attempts to commit suicide
- o
- new or worse depression
- o
- new or worse anxiety
- o
- feeling agitated or restless
- o
- panic attacks
- o
- trouble sleeping (insomnia)
- o
- new or worse irritability
- o
- acting aggressive, being angry, or violent
- o
- acting on dangerous impulses
- o
- an extreme increase in activity and talking (mania)
- o
- other unusual changes in behavior or mood
- How can I watch for early symptoms of suicidal thoughts and actions?
- o
- Pay attention to any changes, especially sudden changes, in mood, behaviors, thoughts, or feelings.
- o
- Keep all follow-up visits with your healthcare provider as scheduled.
- Call your healthcare provider between visits as needed, especially if you are worried about symptoms.
- Do not stop CELONTIN without first talking to a healthcare provider.
- Stopping CELONTIN suddenly can cause serious problems. Stopping a seizure medicine suddenly in a patient who has epilepsy can cause seizures that will not stop (status epilepticus).
- Suicidal thoughts or actions can be caused by things other than medicines. If you have suicidal thoughts or actions, your healthcare provider may check for other causes.
What is CELONTIN?
What is CELONTIN?
CELONTIN is a prescription medicine used to treat absence (petit mal) seizures that have not gotten better with other seizure medicines.
Who should not take CELONTIN?
Who should not take CELONTIN?
Do not take CELONTIN if you are allergic to succinimides (methsuximide or ethosuximide) or any of the ingredients in CELONTIN. See the end of this Medication Guide for a complete list of ingredients in CELONTIN.
What should I tell my healthcare provider before taking CELONTIN?
What should I tell my healthcare provider before taking CELONTIN?
Before you take CELONTIN, tell your healthcare provider if you:
- •
- have or have had liver problems
- •
- have or have had depression, mood problems or suicidal thoughts or behavior
- •
- have any other medical conditions
- •
- are pregnant or plan to become pregnant. It is not known if CELONTIN can harm your unborn baby. Tell your healthcare provider right away if you become pregnant while taking CELONTIN. You and your healthcare provider will decide if you should take CELONTIN while you are pregnant.
- o
- If you become pregnant while taking CELONTIN, talk to your healthcare provider about registering with the North American Antiepileptic Drug (NAAED) Pregnancy Registry. The purpose of this registry is to collect information about the safety of antiepileptic drugs during pregnancy. You can enroll in this registry by calling 1‑888‑233‑2334.
- •
- are breastfeeding or plan to breastfeed. It is not known if CELONTIN can pass into breast milk. You and your healthcare provider should decide how you will feed your baby while you take CELONTIN.
Tell your healthcare provider about all the medicines you take, including prescription and non-prescription medicines, vitamins, and herbal supplements. Taking CELONTIN with certain other medicines can cause side effects or affect how well they work. Do not start or stop other medicines without talking to your healthcare provider.
Know the medicines you take. Keep a list of them with you to show your healthcare provider and pharmacist each time you get a new medicine.
How should I take CELONTIN?
How should I take CELONTIN?
- •
- Take CELONTIN exactly as prescribed. Your healthcare provider will tell you how much CELONTIN to take.
- •
- Your healthcare provider may change your dose. Do not change your dose of CELONTIN without talking to your healthcare provider.
- •
- If you take too much CELONTIN, call your healthcare provider or your local Poison Control Center right away.
What should I avoid while taking CELONTIN?
What should I avoid while taking CELONTIN?
- •
- Do not drink alcohol or take other medicines that make you sleepy or dizzy while taking CELONTIN without first discussing this with your healthcare provider. CELONTIN taken with alcohol or medicines that cause sleepiness or dizziness may make your sleepiness or dizziness worse.
- •
- Do not drive, operate heavy machinery, or do other dangerous activities until you know how CELONTIN affects you. CELONTIN can slow your thinking and motor skills.
What are the possible side effects of CELONTIN?
What are the possible side effects of CELONTIN?
- •
- See "What is the most important information I should know about CELONTIN?"
CELONTIN may cause other serious side effects, including:
- •
- Grand mal seizures can happen more often or become worse
The most common side effects of CELONTIN include:
|
|
Tell your healthcare provider about any side effect that bothers you or that does not go away.
These are not all the possible side effects with CELONTIN. For more information, ask your healthcare provider or pharmacist.
Call your doctor for medical advice about side effects. You may report side effects to FDA at 1‑800‑FDA‑1088.
How should I store CELONTIN?
How should I store CELONTIN?
- •
- Store CELONTIN at room temperature, between 59°F to 86°F (15°C to 30°C).
- •
- Keep CELONTIN capsules in a dry place.
- •
- Keep CELONTIN out of the light.
- •
- Protect CELONTIN from heat.
- •
- Do not use CELONTIN capsules that if they do not look full or if the contents have melted.
Keep CELONTIN and all medicines out of the reach of children.
General information about CELONTIN
General information about CELONTIN
Medicines are sometimes prescribed for purposes other than those listed in a Medication Guide. Do not use CELONTIN for a condition for which it was not prescribed. Do not give CELONTIN to other people, even if they have the same condition. It may harm them.
This Medication Guide summarizes the most important information about CELONTIN. If you would like more information, talk with your healthcare provider. You can ask your healthcare provider or pharmacist for information about CELONTIN that is written for healthcare professionals.
For more information, go to www.pfizer.com or call 1-800-438-1985.
What are the ingredients in CELONTIN?
What are the ingredients in CELONTIN?
Active ingredient: methsuximide
Inactive ingredients: starch, colloidal silicon dioxide NF, D&C yellow No. 10, FD&C yellow No.6, gelatin NF, and sodium lauryl sulfate NF.
This Medication Guide has been approved by the U.S. Food and Drug Administration.
LAB-0404-3.0
Revised March 2023
Full Patient Information
Full Patient Information
MEDICATION GUIDE
CELONTIN (Suh lŏn' tĭn)
(methsuximide)
Capsules
Read this Medication Guide before you start taking CELONTIN and each time you get a refill. There may be new information. This information does not take the place of talking to your healthcare provider about your medical condition or treatment. If you have any questions about CELONTIN, ask your healthcare provider or pharmacist.
What is the most important information I should know about CELONTIN?
Do not stop taking CELONTIN without first talking to your healthcare provider. Stopping CELONTIN suddenly can cause serious problems.
CELONTIN can cause serious side effects, including:
- 1.
- Rare but serious blood problems that may be life-threatening. Call your healthcare provider right away if you have:
- o
- fever, swollen glands, or sore throat that come and go or do not go away
- o
- frequent infections or an infection that does not go away
- o
- easy bruising
- o
- red or purple spots on your body
- o
- bleeding gums or nose bleeds
- o
- severe fatigue or weakness
- 2.
- Systematic Lupus Erythematosus. Call your healthcare provider right away if you have any of these symptoms:
- o
- joint pain and swelling
- o
- muscle pain
- o
- fatigue
- o
- low-grade fever
- o
- pain in the chest that is worse with breathing
- o
- unexplained skin rash
- 3.
- Like other antiepileptic drugs, CELONTIN may cause suicidal thoughts or actions in a very small number of people, about 1 in 500.
Call a healthcare provider right away if you have any of these symptoms, especially if they are new, worse, or worry you:
- o
- thoughts about suicide or dying
- o
- attempts to commit suicide
- o
- new or worse depression
- o
- new or worse anxiety
- o
- feeling agitated or restless
- o
- panic attacks
- o
- trouble sleeping (insomnia)
- o
- new or worse irritability
- o
- acting aggressive, being angry, or violent
- o
- acting on dangerous impulses
- o
- an extreme increase in activity and talking (mania)
- o
- other unusual changes in behavior or mood
- How can I watch for early symptoms of suicidal thoughts and actions?
- o
- Pay attention to any changes, especially sudden changes, in mood, behaviors, thoughts, or feelings.
- o
- Keep all follow-up visits with your healthcare provider as scheduled.
- Call your healthcare provider between visits as needed, especially if you are worried about symptoms.
- Do not stop CELONTIN without first talking to a healthcare provider.
- Stopping CELONTIN suddenly can cause serious problems. Stopping a seizure medicine suddenly in a patient who has epilepsy can cause seizures that will not stop (status epilepticus).
- Suicidal thoughts or actions can be caused by things other than medicines. If you have suicidal thoughts or actions, your healthcare provider may check for other causes.
What is CELONTIN?
CELONTIN is a prescription medicine used to treat absence (petit mal) seizures that have not gotten better with other seizure medicines.
Who should not take CELONTIN?
Do not take CELONTIN if you are allergic to succinimides (methsuximide or ethosuximide) or any of the ingredients in CELONTIN. See the end of this Medication Guide for a complete list of ingredients in CELONTIN.
What should I tell my healthcare provider before taking CELONTIN?
Before you take CELONTIN, tell your healthcare provider if you:
- •
- have or have had liver problems
- •
- have or have had depression, mood problems or suicidal thoughts or behavior
- •
- have any other medical conditions
- •
- are pregnant or plan to become pregnant. It is not known if CELONTIN can harm your unborn baby. Tell your healthcare provider right away if you become pregnant while taking CELONTIN. You and your healthcare provider will decide if you should take CELONTIN while you are pregnant.
- o
- If you become pregnant while taking CELONTIN, talk to your healthcare provider about registering with the North American Antiepileptic Drug (NAAED) Pregnancy Registry. The purpose of this registry is to collect information about the safety of antiepileptic drugs during pregnancy. You can enroll in this registry by calling 1‑888‑233‑2334.
- •
- are breastfeeding or plan to breastfeed. It is not known if CELONTIN can pass into breast milk. You and your healthcare provider should decide how you will feed your baby while you take CELONTIN.
Tell your healthcare provider about all the medicines you take, including prescription and non-prescription medicines, vitamins, and herbal supplements. Taking CELONTIN with certain other medicines can cause side effects or affect how well they work. Do not start or stop other medicines without talking to your healthcare provider.
Know the medicines you take. Keep a list of them with you to show your healthcare provider and pharmacist each time you get a new medicine.
How should I take CELONTIN?
- •
- Take CELONTIN exactly as prescribed. Your healthcare provider will tell you how much CELONTIN to take.
- •
- Your healthcare provider may change your dose. Do not change your dose of CELONTIN without talking to your healthcare provider.
- •
- If you take too much CELONTIN, call your healthcare provider or your local Poison Control Center right away.
What should I avoid while taking CELONTIN?
- •
- Do not drink alcohol or take other medicines that make you sleepy or dizzy while taking CELONTIN without first discussing this with your healthcare provider. CELONTIN taken with alcohol or medicines that cause sleepiness or dizziness may make your sleepiness or dizziness worse.
- •
- Do not drive, operate heavy machinery, or do other dangerous activities until you know how CELONTIN affects you. CELONTIN can slow your thinking and motor skills.
What are the possible side effects of CELONTIN?
- •
- See "What is the most important information I should know about CELONTIN?"
CELONTIN may cause other serious side effects, including:
- •
- Grand mal seizures can happen more often or become worse
The most common side effects of CELONTIN include:
|
|
Tell your healthcare provider about any side effect that bothers you or that does not go away.
These are not all the possible side effects with CELONTIN. For more information, ask your healthcare provider or pharmacist.
Call your doctor for medical advice about side effects. You may report side effects to FDA at 1‑800‑FDA‑1088.
How should I store CELONTIN?
- •
- Store CELONTIN at room temperature, between 59°F to 86°F (15°C to 30°C).
- •
- Keep CELONTIN capsules in a dry place.
- •
- Keep CELONTIN out of the light.
- •
- Protect CELONTIN from heat.
- •
- Do not use CELONTIN capsules that if they do not look full or if the contents have melted.
Keep CELONTIN and all medicines out of the reach of children.
General information about CELONTIN
Medicines are sometimes prescribed for purposes other than those listed in a Medication Guide. Do not use CELONTIN for a condition for which it was not prescribed. Do not give CELONTIN to other people, even if they have the same condition. It may harm them.
This Medication Guide summarizes the most important information about CELONTIN. If you would like more information, talk with your healthcare provider. You can ask your healthcare provider or pharmacist for information about CELONTIN that is written for healthcare professionals.
For more information, go to www.pfizer.com or call 1-800-438-1985.
What are the ingredients in CELONTIN?
Active ingredient: methsuximide
Inactive ingredients: starch, colloidal silicon dioxide NF, D&C yellow No. 10, FD&C yellow No.6, gelatin NF, and sodium lauryl sulfate NF.
This Medication Guide has been approved by the U.S. Food and Drug Administration.
LAB-0404-3.0
Revised March 2023
Resources
Didn’t find what you were looking for?
Contact us.
Chat online with Pfizer Medical Information regarding your inquiry on a Pfizer medicine.
*Speak with a Pfizer Medical Information Professional regarding your medical inquiry. Available 9AM-5PM ET Monday to Friday; excluding holidays.
Submit a medical question for Pfizer prescription products.
Report Adverse Event
Pfizer Safety
To report an adverse event related to a Pfizer product and you are not part of a clinical trial* for this product, click the link below to submit your information: Pfizer Safety Reporting Site
*If you are involved in a clinical trial for either product, adverse events should be reported to your coordinating study site.
If you cannot use the above website to report an adverse event related to a Pfizer product, please call Pfizer Medical Information at (800) 438-1985.
FDA Medwatch
You may also contact the U.S. Food and Drug Administration (FDA) directly to report adverse events or product quality concerns either online at www.fda.gov/medwatch or by calling (800)-332-1088.