TUKYSA® Description
(tucatinib)
11 DESCRIPTION
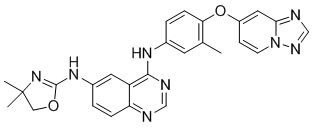
Tucatinib is a kinase inhibitor. The chemical name is (N4-(4-([1,2,4]triazolo[1,5-a]pyridin-7-yloxy)-3-methylphenyl)-N6-(4,4-dimethyl-4,5-dihydrooxazol-2-yl)quinazoline-4,6-diamine. The molecular formula is C26H24N8O2 and the molecular weight is 480.52 g/mol. The chemical structure is as follows:
TUKYSA (tucatinib) is supplied as 50 mg and 150 mg film-coated tablets for oral use and contain the following inactive ingredients:
Tablet core: copovidone, crospovidone, sodium chloride, potassium chloride, sodium bicarbonate, colloidal silicon dioxide, magnesium stearate, and microcrystalline cellulose.
Coating: yellow film coat: polyvinyl alcohol, titanium dioxide, macrogol/polyethylene glycol, talc, and yellow iron oxide non-irradiated.
Each TUKYSA 50 mg tablet contains 10.10 mg (0.258 mEq) potassium and 9.21 mg (0.401 mEq) sodium.
Each TUKYSA 150 mg tablet contains 30.29 mg (0.775 mEq) potassium and 27.64 mg (1.202 mEq) sodium.
Find TUKYSA® medical information:

Find TUKYSA® medical information:
TUKYSA® Quick Finder
Health Professional Information
Description
11 DESCRIPTION
Tucatinib is a kinase inhibitor. The chemical name is (N4-(4-([1,2,4]triazolo[1,5-a]pyridin-7-yloxy)-3-methylphenyl)-N6-(4,4-dimethyl-4,5-dihydrooxazol-2-yl)quinazoline-4,6-diamine. The molecular formula is C26H24N8O2 and the molecular weight is 480.52 g/mol. The chemical structure is as follows:
TUKYSA (tucatinib) is supplied as 50 mg and 150 mg film-coated tablets for oral use and contain the following inactive ingredients:
Tablet core: copovidone, crospovidone, sodium chloride, potassium chloride, sodium bicarbonate, colloidal silicon dioxide, magnesium stearate, and microcrystalline cellulose.
Coating: yellow film coat: polyvinyl alcohol, titanium dioxide, macrogol/polyethylene glycol, talc, and yellow iron oxide non-irradiated.
Each TUKYSA 50 mg tablet contains 10.10 mg (0.258 mEq) potassium and 9.21 mg (0.401 mEq) sodium.
Each TUKYSA 150 mg tablet contains 30.29 mg (0.775 mEq) potassium and 27.64 mg (1.202 mEq) sodium.
Health Professional Information
{{section_name_patient}}
{{section_body_html_patient}}
Resources
Didn’t find what you were looking for? Contact us.
Chat online with Pfizer Medical Information regarding your inquiry on a Pfizer medicine.
*Speak with a Pfizer Medical Information Professional regarding your medical inquiry. Available 9AM-5PM ET Monday to Friday; excluding holidays.
Submit a medical question for Pfizer prescription products.
Report Adverse Event
Pfizer Safety
To report an adverse event related to the Pfizer-BioNTech COVID-19 Vaccine, and you are not part of a clinical trial* for this product, click the link below to submit your information:
Pfizer Safety Reporting Site*If you are involved in a clinical trial for this product, adverse events should be reported to your coordinating study site.
If you cannot use the above website, or would like to report an adverse event related to a different Pfizer product, please call Pfizer Safety at (800) 438-1985.
FDA Medwatch
You may also contact the U.S. Food and Drug Administration (FDA) directly to report adverse events or product quality concerns either online at www.fda.gov/medwatch or call (800) 822-7967.